Balanced Equation For Combustion Of Methane
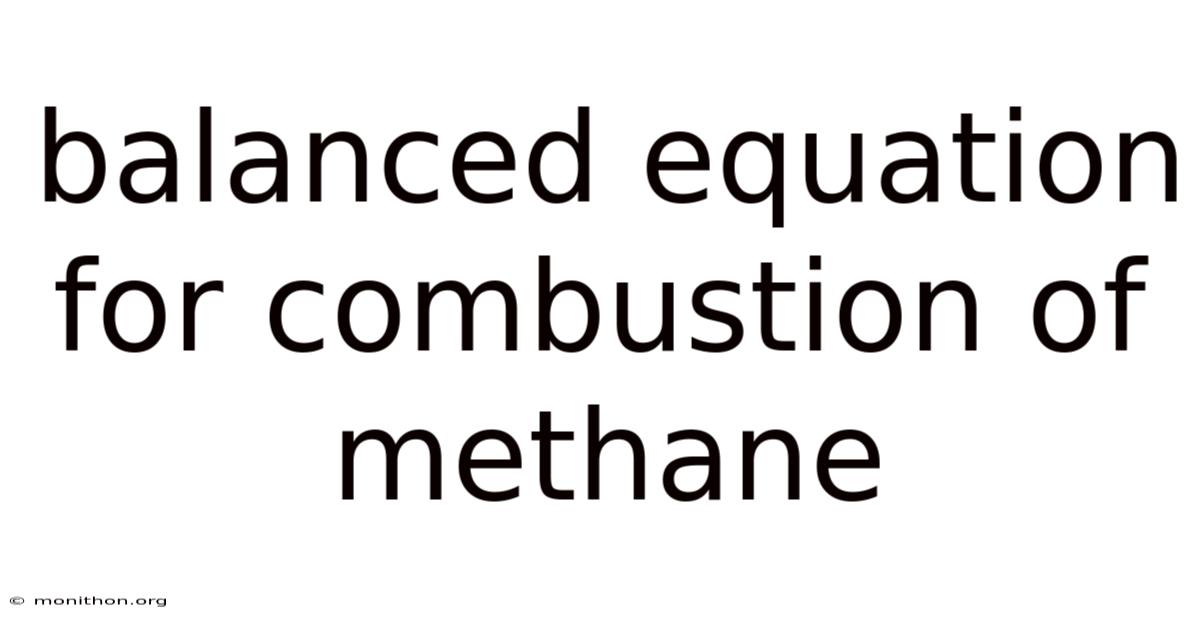
Balanced Equation for Combustion of Methane: A Comprehensive Guide
Methane (CH₄), the simplest hydrocarbon and the primary component of natural gas, plays a pivotal role in energy production and environmental science. Its combustion—a reaction with oxygen (O₂)—is a cornerstone of industrial processes, heating systems, and even rocket propulsion. However, understanding the balanced chemical equation for this reaction is essential for optimizing energy efficiency, minimizing environmental impact, and ensuring safety. This article delves into the balanced equation for methane combustion, its derivation, scientific significance, and real-world applications.
Introduction
Combustion reactions are exothermic processes where a substance reacts with oxygen to produce heat and light. Methane combustion is particularly notable for its role in generating energy, as seen in gas stoves, power plants, and internal combustion engines. The balanced equation for this reaction ensures that the number of atoms of each element remains constant on both sides, adhering to the law of conservation of mass.
The unbalanced equation for methane combustion is:
CH₄ + O₂ → CO₂ + H₂O
Balancing this equation requires adjusting coefficients to equalize the atoms of carbon (C), hydrogen (H), and oxygen (O) on both sides. Let’s break down the process step by step.
Step-by-Step Balancing of the Methane Combustion Equation
Step 1: Write the Unbalanced Equation
Start with the reactants (methane and oxygen) and products (carbon dioxide and water):
CH₄ + O₂ → CO₂ + H₂O
Step 2: Balance Carbon Atoms
There is 1 carbon atom in CH₄ and 1 in CO₂. Carbon is already balanced.
Step 3: Balance Hydrogen Atoms
Methane has 4 hydrogen atoms, while water (H₂O) has 2. To balance hydrogen, place a coefficient of 2 in front of H₂O:
CH₄ + O₂ → CO₂ + 2H₂O
Step 4: Balance Oxygen Atoms
On the product side, there are 2 oxygen atoms in CO₂ and 2 × 1 = 2 in H₂O, totaling 4 oxygen atoms. Since O₂ is diatomic, divide the total oxygen atoms by 2 to find the coefficient for O₂:
4 oxygen atoms ÷ 2 = 2 O₂ molecules
Update the equation:
CH₄ + 2O₂ → CO₂ + 2H₂O
Step 5: Verify the Balance
- Carbon: 1 on both sides ✅
- Hydrogen: 4 on both sides ✅
- Oxygen: (2 × 2) = 4 on the left; (2 + 2 × 1) = 4 on the right ✅
The balanced equation is:
CH₄ + 2O₂ → CO₂ + 2H₂O
Scientific Explanation of Methane Combustion
1. Exothermic Nature
Methane combustion releases energy (heat and light) because the bonds in O₂ and CH₄ are broken, and new, stronger bonds in CO₂ and H₂O are formed. The enthalpy change (ΔH) for this reaction is approximately -890 kJ/mol, making it highly exothermic.
2. Role of Activation Energy
For combustion to occur, methane and oxygen must overcome an activation energy barrier. In industrial settings, this is achieved via sparks, flames, or catalysts. In natural gas stoves, a pilot light provides the initial energy to initiate the reaction.
3. Thermodynamic Favorability
The reaction is thermodynamically favorable due to the stability of CO₂ and H₂O. The products have lower potential energy than the reactants, releasing energy to the surroundings.
4. Environmental Impact
While methane combustion is efficient, it produces CO₂, a greenhouse gas contributing to global warming. Additionally, incomplete combustion can generate carbon monoxide (CO), a toxic gas, and soot, which contributes to air pollution.
Applications of Methane Combustion
1. Energy Production
Methane is burned in gas turbines, boilers, and internal combustion engines to generate electricity and mechanical power. Its high energy density (55.7 MJ/kg) makes it a preferred fuel for heating and transportation.
2. Industrial Processes
- Steel Manufacturing: Methane powers furnaces for smelting iron ore.
- Chemical Synthesis: It serves as a feedstock for producing methanol, ammonia, and other chemicals.
3. Everyday Use
Natural gas (primarily methane) is used in residential heating, cooking, and electricity generation. Its clean-burning properties compared to coal or oil reduce particulate emissions.
FAQ: Common Questions About Methane Combustion
Q1: Why is balancing the equation important?
Balancing ensures the law of conservation of mass is upheld. Unbalanced equations could lead to incorrect stoichiometric calculations in industrial processes or environmental modeling.
Q2: What happens if oxygen is limited during combustion?
Insufficient oxygen leads to incomplete combustion, producing carbon monoxide (CO), soot (carbon particles), and reduced energy output. This is why proper ventilation is critical in gas appliances.
Q3: Can methane combustion be made more efficient?
Yes! Catalytic converters and advanced combustion technologies improve efficiency by ensuring complete oxidation of methane, minimizing waste products.
Q4: Is methane a renewable resource?
No. Methane from natural gas is a fossil fuel. However, biogas (methane from organic waste) is renewable and considered carbon-neutral if sustainably managed.
Conclusion
The balanced equation
Conclusion:The Balanced Equation and Beyond
The balanced equation for methane combustion, CH₄ + 2O₂ → CO₂ + 2H₂O, is far more than a mere mathematical exercise. It is the fundamental blueprint that governs the entire process, dictating the precise quantities of fuel and oxidant required for complete combustion, the exact energy released, and the nature of the waste products formed. This stoichiometric precision is not just academic; it is the cornerstone of efficiency, safety, and environmental responsibility in every application where methane is burned.
From the controlled ignition of a pilot light in a home stove to the massive turbines generating electricity for cities, understanding and applying this balanced equation ensures optimal energy extraction, minimizes harmful byproducts like carbon monoxide and soot, and allows for the effective design and operation of combustion systems. It underpins the thermodynamic favorability that makes methane a valuable energy source, while simultaneously highlighting the environmental challenge posed by its primary combustion product, carbon dioxide.
Therefore, mastering the balanced equation is not an end in itself, but the essential first step towards harnessing the power of methane combustion responsibly. It provides the critical foundation upon which we build efficient energy systems, develop cleaner technologies, and strive to mitigate the environmental impact of this ubiquitous fuel. The equation reveals the inherent potential and inherent risks of methane combustion, demanding careful management to maximize benefits while minimizing harm.
Conclusion: The Balanced Equation and Beyond
The balanced equation for methane combustion, CH₄ + 2O₂ → CO₂ + 2H₂O, is far more than a mere mathematical exercise. It is the fundamental blueprint that governs the entire process, dictating the precise quantities of fuel and oxidant required for complete combustion, the exact energy released, and the nature of the waste products formed. This stoichiometric precision is not just academic; it is the cornerstone of efficiency, safety, and environmental responsibility in every application where methane is burned.
From the controlled ignition of a pilot light in a home stove to the massive turbines generating electricity for cities, understanding and applying this balanced equation ensures optimal energy extraction, minimizes harmful byproducts like carbon monoxide and soot, and allows for the effective design and operation of combustion systems. It underpins the thermodynamic favorability that makes methane a valuable energy source, while simultaneously highlighting the environmental challenge posed by its primary combustion product, carbon dioxide.
Therefore, mastering the balanced equation is not an end in itself, but the essential first step towards harnessing the power of methane combustion responsibly. It provides the critical foundation upon which we build efficient energy systems, develop cleaner technologies, and strive to mitigate the environmental impact of this ubiquitous fuel. The equation reveals the inherent potential and inherent risks of methane combustion, demanding careful management to maximize benefits while minimizing harm.
In conclusion, the balanced equation for methane combustion is a powerful tool that holds the key to unlocking the full potential of this energy source while minimizing its environmental footprint. By understanding and applying this fundamental principle, we can create a more sustainable future, where energy is harnessed efficiently, safely, and responsibly. The balanced equation is not just a mathematical concept; it is a blueprint for a brighter, more sustainable tomorrow.
Latest Posts
Latest Posts
-
250 Mg Is How Many Ml
Mar 20, 2026
-
How Do You Combine Like Terms
Mar 20, 2026
-
How Did Leonardo Da Vinci Impact The Renaissance
Mar 20, 2026
-
8 1 3 As A Fraction
Mar 20, 2026
-
How Do You Say Thank You In Arabic Language
Mar 20, 2026