Chemical Equation For Vinegar And Baking Soda
monithon
Mar 10, 2026 · 7 min read
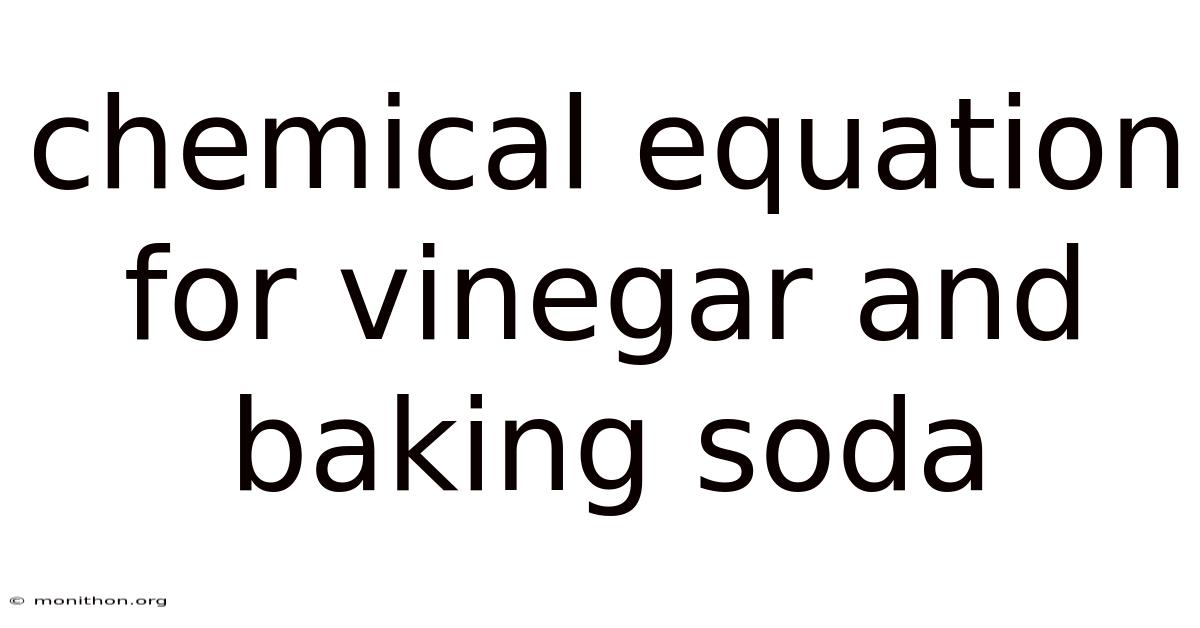
Table of Contents
Understanding the chemical reaction between vinegar and baking soda is a fascinating example of how everyday substances interact in a simple yet powerful way. This reaction not only produces a delightful fizz but also demonstrates the principles of chemistry in action. Whether you're a student exploring science or a curious learner, this article will break down the process, highlight its significance, and help you grasp the science behind this classic experiment.
The chemical equation for vinegar and baking soda is a simple yet profound illustration of an acid-base reaction. When these two common household items come together, they undergo a transformation that results in the release of carbon dioxide gas. This reaction is not only visually engaging but also scientifically meaningful, making it a great subject for educational purposes.
To begin, it’s essential to understand what vinegar and baking soda are. Vinegar is a common household liquid made primarily from acetic acid, while baking soda is a white, powdery substance known as sodium bicarbonate. When these two substances are mixed, they react in a way that produces a bubbly effect, which many people find exciting. This reaction is a prime example of a neutralization reaction, where an acid and a base combine to form a salt and water.
The process starts when the acidic vinegar comes into contact with the baking soda. The acetic acid in the vinegar reacts with the sodium bicarbonate in the baking soda. This interaction leads to the formation of new compounds, including carbon dioxide gas, water, and sodium acetate. The carbon dioxide gas is what causes the fizzing sound and the visible bubbles.
What makes this reaction particularly interesting is the role of temperature in the process. When the two substances are mixed, the reaction becomes more vigorous. However, the rate of the reaction can be influenced by factors such as the concentration of the substances and the surface area of contact. For instance, if you crush the baking soda into smaller pieces, it will react faster because it increases the exposure of the baking soda to the vinegar.
The importance of this reaction extends beyond just a fun experiment. It helps us understand basic chemical principles that are foundational in many areas of science. By observing this reaction, students can grasp concepts like acid-base chemistry, gas production, and chemical changes. These ideas are crucial for more advanced studies in chemistry and even in everyday applications like cooking or cleaning.
To delve deeper into the science behind this reaction, let’s break it down step by step. When you mix vinegar and baking soda, the acetic acid in the vinegar reacts with the sodium bicarbonate in the baking soda. This reaction can be represented by the following chemical equation:
Acetic acid + Sodium bicarbonate → Sodium acetate + Water + Carbon dioxide gas
This equation shows the transformation of the original substances into new products. The sodium acetate is a salt that remains in the solution, while water is produced as a byproduct. The carbon dioxide gas is what creates the bubbles, making the reaction visually appealing.
It’s important to note that this reaction is exothermic, meaning it releases heat. This is why you might feel a slight warmth when the mixture is stirred. The heat generated is a result of the energy released during the chemical transformation.
For those interested in the real-world applications, this reaction is not just limited to the classroom. It plays a role in various industries. For example, baking soda is used in food preservation and as a leavening agent, while vinegar is a common ingredient in cleaning products and cooking. Understanding how these substances interact can help in developing new products or improving existing ones.
Moreover, this reaction is a great way to teach safety practices. When handling vinegar and baking soda, it’s crucial to wear protective gear such as gloves and goggles. This not only protects the user but also emphasizes the importance of safety protocols in scientific experiments.
Another key point to consider is the environmental impact of this reaction. While the process itself is harmless, the production of vinegar and baking soda can have ecological effects. For instance, if vinegar is made from natural ingredients, it’s more sustainable than synthetic alternatives. This connection between chemistry and sustainability highlights the relevance of understanding these reactions in a broader context.
The visual appeal of the reaction also makes it a popular choice for science demonstrations. Teachers often use this experiment to engage students, as the bubbling and fizzing make it an exciting visual experience. This not only enhances learning but also encourages curiosity about the world of science.
In addition to its educational value, this reaction has historical significance. The use of vinegar and baking soda dates back centuries, with ancient civilizations using these substances for various purposes. From preserving food to cleaning, their applications have evolved over time. Learning about this history adds a layer of context to the scientific process.
When exploring this topic further, it’s worth considering the variations in the reaction. For example, if you use a stronger vinegar or more baking soda, the reaction becomes more intense. This variation can be used to teach experimental design and control variables—key concepts in scientific inquiry.
Understanding the chemical equation also helps in identifying other reactions that involve similar components. For instance, other acidic substances like lemon juice or citrus fruits can also react with baking soda, producing carbon dioxide. This connection reinforces the interconnectedness of chemical knowledge.
The benefits of this article extend beyond just explaining the reaction. It encourages readers to think critically about chemical processes and their real-life implications. By breaking down the steps and explaining the why behind each action, we empower learners to apply their knowledge in practical situations.
Moreover, this topic is relevant to many areas of life. Whether you’re a student preparing for a science fair, a teacher looking for a lesson plan, or a homeowner interested in DIY projects, this article provides valuable insights. It’s a reminder that science is not just about formulas but about understanding the world around us.
In conclusion, the chemical equation for vinegar and baking soda is more than just a simple reaction—it’s a gateway to understanding the wonders of chemistry. By exploring this process, we not only learn about acid-base interactions but also develop a deeper appreciation for the science that shapes our daily lives. Whether you’re curious about how things work or why certain reactions happen, this article offers a clear and engaging exploration of this classic experiment.
Remember, science is all about curiosity and learning. The next time you see a fizzing mixture, take a moment to appreciate the chemistry at play. This small reaction is a perfect example of how knowledge can transform simple observations into meaningful discoveries. Let this article inspire you to dive deeper into the fascinating world of science.
…and ultimately, it’s a testament to the power of observation and experimentation – a cornerstone of scientific progress. Beyond the immediate visual spectacle of the bubbling mixture, this seemingly basic reaction embodies fundamental principles applicable across a vast spectrum of scientific disciplines.
Consider, for instance, the broader implications of carbon dioxide production. This reaction is a microcosm of larger processes occurring within our atmosphere, influencing climate change and the delicate balance of ecosystems. Similarly, the principles of acid-base reactions underpin countless industrial processes, from the production of fertilizers to the manufacturing of pharmaceuticals.
Furthermore, this activity provides a tangible entry point for exploring concepts like stoichiometry – the quantitative relationship between reactants and products – and conservation of mass. Students can readily manipulate the quantities of vinegar and baking soda to observe the direct correlation between input and output, solidifying these crucial mathematical skills.
Finally, let’s not underestimate the value of this experiment as a springboard for fostering a genuine scientific mindset. It encourages students to formulate hypotheses, design experiments, collect and analyze data, and draw conclusions – skills that are invaluable not just in the laboratory, but in navigating the complexities of the world around them.
Therefore, the vinegar and baking soda reaction, often dismissed as a simple demonstration, represents a surprisingly rich and multifaceted learning opportunity. It’s a reminder that even the most commonplace phenomena can hold profound scientific significance, and that a curious mind, coupled with careful observation, can unlock a universe of understanding.
Latest Posts
Latest Posts
-
Which Statement Is Correct About A Sample Of Liquid Water
Mar 10, 2026
-
Advantages And Disadvantages Of Sexual Reproduction
Mar 10, 2026
-
7am To 7pm Is How Many Hours
Mar 10, 2026
-
Whats The Difference Between Romanticism And Transcendentalism
Mar 10, 2026
-
How Many Oz In A 1 4 Pound
Mar 10, 2026
Related Post
Thank you for visiting our website which covers about Chemical Equation For Vinegar And Baking Soda . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.