Ethanol And Oxygen Reaction Acetic Acid Balanced Equation
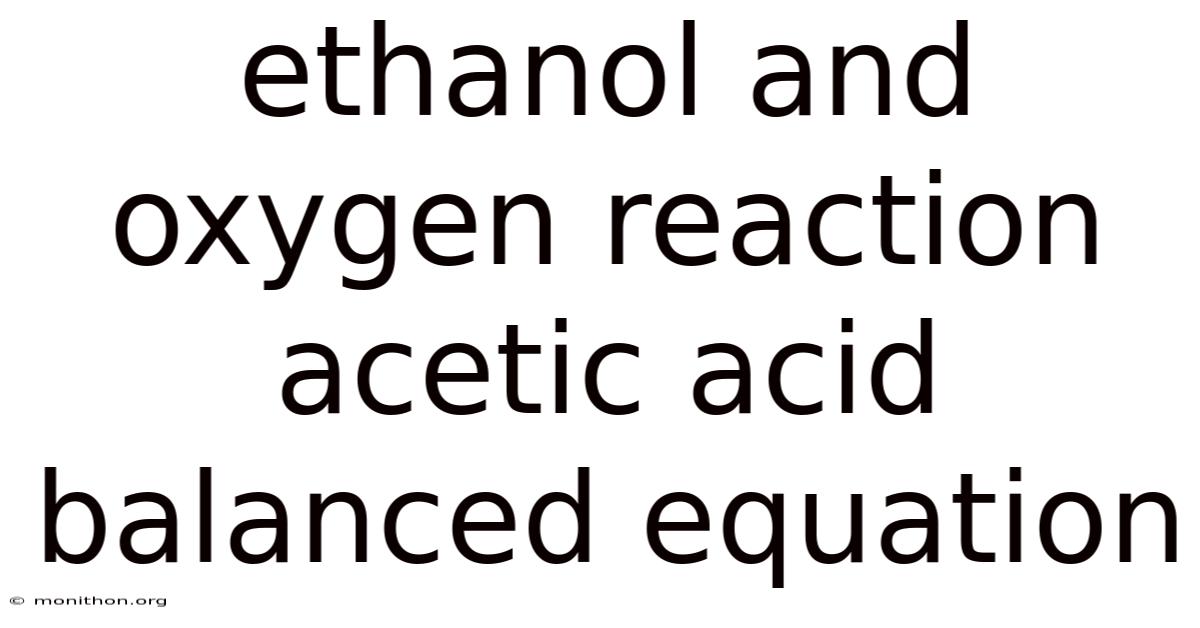
The combustion of ethanol(C₂H₅OH) with oxygen (O₂) is a fundamental chemical reaction, producing carbon dioxide and water. This process is central to understanding fuel combustion and energy release. The balanced equation for this reaction is:
C₂H₅OH + 3O₂ → 2CO₂ + 3H₂O
This equation signifies that one molecule of ethanol reacts with three molecules of oxygen gas to yield two molecules of carbon dioxide and three molecules of water. This reaction releases significant energy, making ethanol a viable fuel source.
Oxidation to Acetic Acid
Ethanol can also undergo oxidation to form acetic acid (CH₃COOH), a key industrial process. This transformation requires an oxidizing agent, typically potassium dichromate (K₂Cr₂O₇) in sulfuric acid (H₂SO₄), under controlled conditions. The balanced equation for this oxidation is:
C₂H₅OH + 2[O] → CH₃COOH + H₂O
Here, the oxidizing agent provides the necessary oxygen atoms (represented as [O]), converting the ethanol's hydroxyl group (-OH) into a carbonyl group (C=O) in acetic acid. This reaction is crucial for producing vinegar and various chemical intermediates.
Scientific Explanation
The combustion reaction releases energy because the bonds in the products (CO₂ and H₂O) are stronger than those in the reactants (C₂H₅OH and O₂). The oxygen atoms in O₂ molecules are highly reactive, readily forming stable bonds with carbon and hydrogen atoms. This exothermic reaction powers engines and industrial processes globally. Conversely, the oxidation to acetic acid involves a controlled redox process where ethanol loses electrons (oxidation) while the oxidizing agent gains electrons (reduction). The reaction mechanism involves initial protonation of ethanol, nucleophilic attack by the oxidizing agent, and subsequent dehydration steps.
Steps for Balanced Equations
- Write the Unbalanced Equation: Start with the correct molecular formulas (C₂H₅OH, O₂, CO₂, H₂O).
- Count Atoms: Determine the number of atoms for each element on both sides.
- Balance Carbon: Adjust coefficients to equalize carbon atoms (e.g., 2CO₂ requires 2C₂H₅OH).
- Balance Hydrogen: Add coefficients to balance hydrogen atoms (e.g., 3H₂O requires 6H atoms).
- Balance Oxygen: Calculate remaining oxygen atoms and adjust coefficients accordingly (e.g., 6O atoms from 2CO₂ and 3H₂O require 3O₂ molecules).
FAQ
Q: Why is the combustion equation written with 3O₂?
A: Three oxygen molecules provide the six oxygen atoms needed to form two CO₂ molecules (each requiring two O atoms) and three H₂O molecules (each requiring one O atom).
Q: Can ethanol be oxidized without an agent?
A: No, pure oxygen alone cannot oxidize ethanol to acetic acid; an oxidizing agent like K₂Cr₂O₇ is essential to facilitate the electron transfer.
Q: Is acetic acid formation reversible?
A: The oxidation is typically irreversible under standard conditions, as acetic acid is a more oxidized product.
Conclusion
The reactions of ethanol with oxygen illustrate core principles of chemical energetics and redox chemistry. Whether driving combustion engines or synthesizing acetic acid, these balanced equations provide the foundation for practical applications and deeper scientific understanding. Mastery of such reactions empowers innovation in energy and chemical manufacturing.
Latest Posts
Latest Posts
-
How To Find The Value Of A Function
Mar 21, 2026
-
Is A Vacuole In Plant And Animal Cells
Mar 21, 2026
-
How To Find The Domain In A Graph
Mar 21, 2026
-
What Is The Electron Configuration Of B
Mar 21, 2026
-
Why Did President Monroe Issue The Monroe Doctrine
Mar 21, 2026