Kg M 3 To Lb In 3
monithon
Mar 12, 2026 · 4 min read
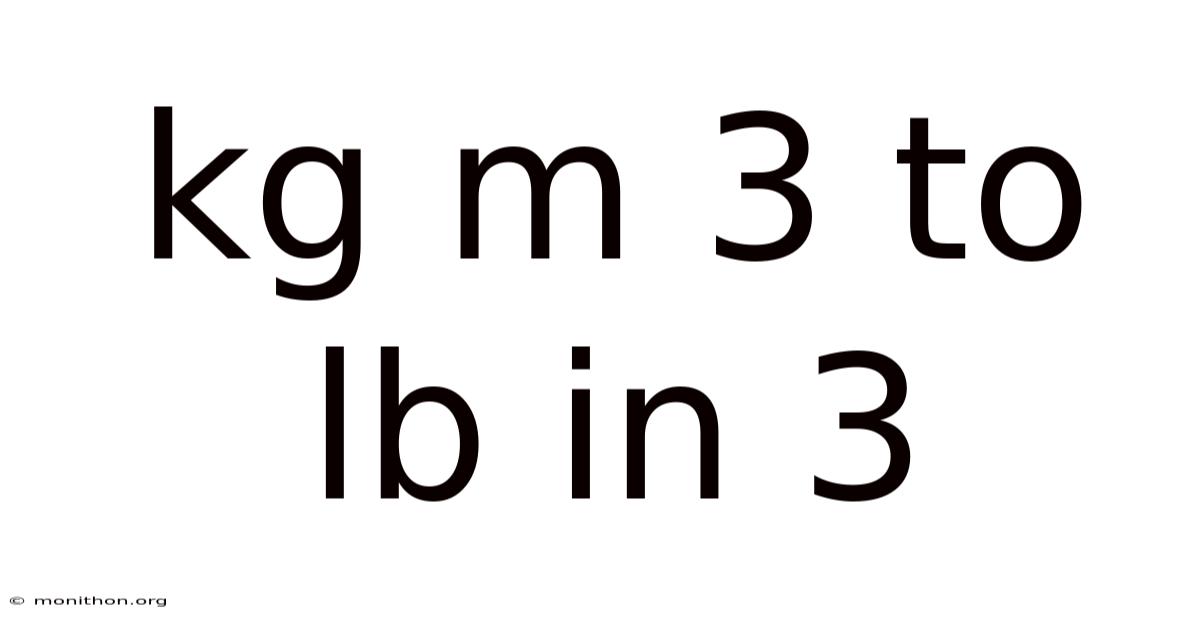
Table of Contents
The concept of unit conversions serves as a cornerstone in scientific inquiry, bridging disparate measurement systems and enabling seamless communication across disciplines. Whether navigating the intricate world of physics, engineering, or everyday life, understanding how quantities relate to one another is paramount. In a realm where precision dictates outcomes, such as in manufacturing, scientific research, or even culinary arts, the ability to translate units accurately ensures reliability and consistency. This article delves deeply into the mechanics behind converting cubic meters to pounds, exploring the underlying principles that underpin such transformations. By examining the interplay between volume, mass, and density, we uncover not only numerical relationships but also the philosophical underpinnings that govern these processes. Such knowledge empowers individuals to apply these principles effectively, whether adjusting industrial processes, conducting experiments, or simply managing household tasks with greater efficiency. The significance of such conversions extends beyond mere calculation; they represent a bridge between abstract concepts and tangible applications, allowing users to translate theoretical knowledge into practical utility. This foundational understanding
To convert a volume expressed in cubic meters (m³) into a weight expressed in pounds (lb), one must first determine the mass of the material occupying that volume. Mass is obtained by multiplying the volume by the substance’s density (ρ), which relates mass to volume through the equation
[ m = V \times \rho . ]
Density is typically reported in kilograms per cubic meter (kg·m⁻³) for solids and liquids, or in grams per liter (g·L⁻¹) for gases under standard conditions. Once the mass in kilograms is known, it is straightforward to convert to pounds using the exact equivalence
[ 1;\text{kg} = 2.20462;\text{lb}. ]
Thus the full conversion chain becomes
[ \text{Weight (lb)} = V;(\text{m}^3) \times \rho;(\text{kg·m}^{-3}) \times 2.20462;(\text{lb·kg}^{-1}). ]
Illustrative Examples | Substance | Typical Density (kg·m⁻³) | Mass of 1 m³ (kg) | Weight of 1 m³ (lb) |
|-----------|--------------------------|-------------------|----------------------| | Water (4 °C) | 999.8 ≈ 1 000 | ≈ 1 000 | ≈ 2 204.6 | | Air (15 °C, 1 atm) | 1.225 | ≈ 1.225 | ≈ 2.70 | | Structural Steel | 7 850 | ≈ 7 850 | ≈ 17 300 | | Ethanol (20 °C) | 789 | ≈ 789 | ≈ 1 740 | | Concrete (typical) | 2 400 | ≈ 2 400 | ≈ 5 291 |
These numbers underscore why the conversion cannot be performed with a universal constant; the material’s density dictates the outcome. For gases, density varies markedly with temperature and pressure, so the ideal‑gas law
[ \rho = \frac{PM}{RT} ]
(where P is pressure, M molar mass, R the universal gas constant, and T absolute temperature) is often employed to compute an appropriate ρ before applying the volume‑to‑weight transformation.
Practical Workflow
- Identify the material and obtain its density under the relevant conditions (tables, material data sheets, or calculations for gases).
- Measure or specify the volume in cubic meters.
- Calculate mass: multiply volume by density.
- Convert mass to pounds: multiply the result by 2.20462.
- Check units to ensure cancellation leaves only pounds.
When high precision is required—such as in aerospace payload calculations or pharmaceutical formulation—consider correcting density for temperature‑induced expansion or compression, and retain sufficient significant figures throughout the computation.
Limitations and Considerations
- Heterogeneous mixtures (e.g., soil, aggregates) may not possess a single, well‑defined density; in such cases, an effective bulk density measured experimentally is preferable.
- Porous materials absorb fluids, altering their apparent density depending on saturation state.
- Non‑Newtonian fluids can exhibit density changes under shear, though for most engineering purposes density remains a reliable scalar property. - Unit consistency is essential; mistakenly using density in g·cm⁻³ without converting to kg·m⁻³ will introduce errors of three orders of magnitude.
By recognizing that weight is not an intrinsic property of volume alone but emerges from the interaction of volume with material density, practitioners can avoid the common pitfall of applying a blanket conversion factor. The method outlined above provides a transparent, reproducible pathway from cubic meters to pounds, adaptable to virtually any substance whose density can be ascertained.
Conclusion
Converting cubic meters to pounds hinges on the fundamental relationship between volume, mass, and density, followed by a straightforward mass‑to‑weight conversion using the kilogram‑to‑pound factor. This process highlights the indispensable role of material‑specific data: without an accurate density value, any volumetric‑to‑gravimetric translation would be meaningless. Whether estimating the load of a concrete slab, sizing a gas storage tank, or formulating a liquid recipe, mastering this conversion empowers scientists, engineers, and technicians to move confidently between abstract measurements and tangible, real‑world outcomes. In a world where precision drives safety, efficiency, and innovation, understanding and correctly applying these principles remains a cornerstone of effective technical practice
Latest Posts
Latest Posts
-
A Rectangle Is Always A Rhombus
Mar 12, 2026
-
What Multiplies To But Adds To
Mar 12, 2026
-
Is 2 3 A Whole Number
Mar 12, 2026
-
What Do All Rhombuses Have In Common
Mar 12, 2026
-
How Do You Say The Answer In Spanish
Mar 12, 2026
Related Post
Thank you for visiting our website which covers about Kg M 3 To Lb In 3 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.