Liquid Octane Reacts With Gaseous Oxygen Gas
monithon
Mar 16, 2026 · 2 min read
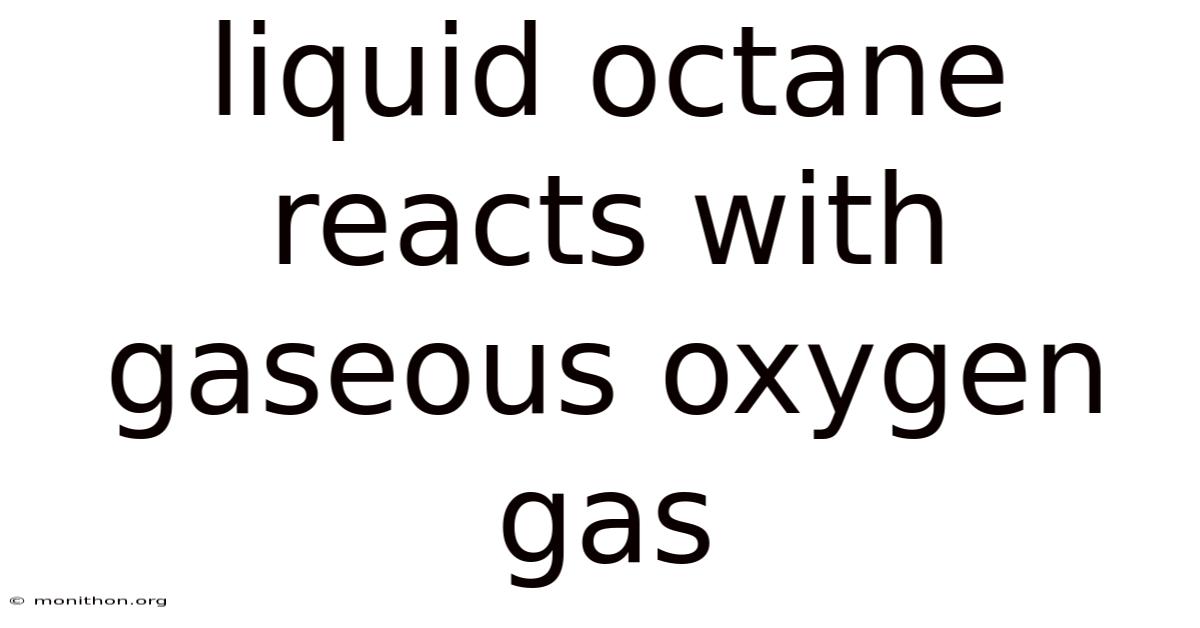
Table of Contents
When liquid octane reacts with gaseous oxygen gas, a combustion reaction occurs that releases a large amount of energy in the form of heat and light. This process is commonly known as burning and is the fundamental principle behind the use of gasoline in engines.
Octane is a hydrocarbon with the molecular formula C₈H₁₈. In its liquid state, it is a major component of gasoline. Oxygen gas, O₂, is a highly reactive molecule found abundantly in the atmosphere. When these two substances come into contact under the right conditions, they undergo a rapid oxidation reaction.
The balanced chemical equation for the complete combustion of octane is:
2 C₈H₁₈ + 25 O₂ → 16 CO₂ + 18 H₂O
This means that two molecules of octane react with twenty-five molecules of oxygen to produce sixteen molecules of carbon dioxide and eighteen molecules of water. The reaction also releases a significant amount of energy, which is why it is exothermic.
For the reaction to begin, an initial energy input is required, usually in the form of a spark or flame. This energy breaks the bonds in the octane and oxygen molecules, allowing new bonds to form in the products. Once the reaction starts, it becomes self-sustaining because the energy released by forming new bonds is greater than the energy required to break the initial bonds.
The energy released during the combustion of octane is measured in terms of enthalpy change, often expressed in kilojoules per mole (kJ/mol). The standard enthalpy of combustion for octane is approximately -5470 kJ/mol, indicating a large release of energy.
This energy release is what makes octane a valuable fuel. In internal combustion engines, the controlled burning of octane-air mixtures produces the force needed to move pistons, which then powers vehicles. The efficiency of this process depends on factors such as the air-to-fuel ratio, temperature, and the presence of catalysts.
Incomplete combustion can occur if there is insufficient oxygen. In such cases, carbon monoxide (CO) and unburned hydrocarbons may form instead of carbon dioxide and water. This not only reduces the energy output but also produces harmful emissions that contribute to air pollution.
The combustion of octane also produces water vapor as a byproduct. While water is not harmful, the carbon dioxide produced is a greenhouse gas that contributes to global warming when released in large quantities from human activities.
Understanding the reaction between liquid octane and gaseous oxygen is crucial for optimizing fuel use, reducing emissions, and developing alternative energy sources. Scientists and engineers continually work on improving combustion efficiency and minimizing environmental impact through better engine designs and cleaner fuels.
In summary, the reaction between liquid octane and gaseous oxygen is a combustion process that releases a large amount of energy. It is the basis for the use of gasoline in engines and has significant implications for energy production and environmental sustainability.
Latest Posts
Latest Posts
-
A Negative Divided By A Positive
Mar 16, 2026
-
How To Find Apothem Of Pentagon
Mar 16, 2026
-
Which Of The Following Is Equivalent To The Expression Above
Mar 16, 2026
-
D Varies Directly With W And Inversely With P
Mar 16, 2026
-
Domain And Range Of Ordered Pairs
Mar 16, 2026
Related Post
Thank you for visiting our website which covers about Liquid Octane Reacts With Gaseous Oxygen Gas . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.