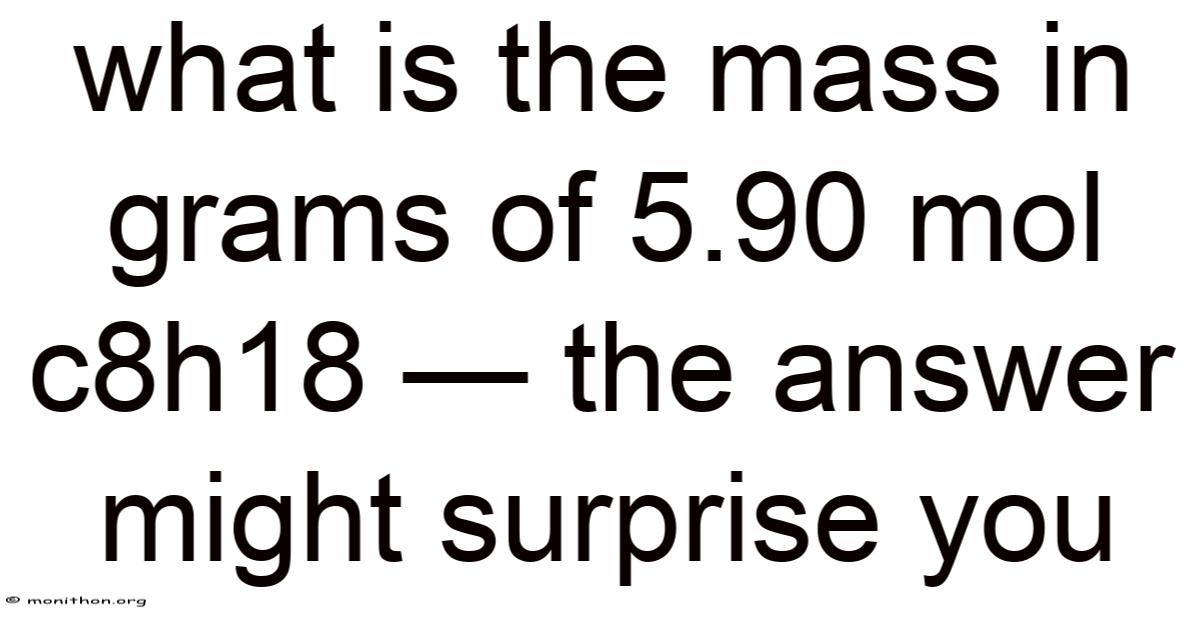What Is the Mass in Grams of 5.90 mol C₈H₁₈?
Ever seen a chemistry problem that looks like a math puzzle and wondered, “How do I actually get that number?” The answer is simple: you just multiply the number of moles by the molar mass. But the trick is knowing how to get that molar mass right and why it matters. Stick with me and you’ll walk away with a clear method, a few handy shortcuts, and a sense of how this tiny calculation fits into the bigger picture of chemical science.
What Is the Mass in Grams of 5.90 mol C₈H₁₈?
In plain terms, you’re asking: “If I have 5.90 moles of octane (C₈H₁₈), how many grams does that weigh?” Octane is the compound that powers gasoline, so this question pops up in everything from fuel efficiency studies to laboratory titrations Nothing fancy..
[ \text{mass (g)} = \text{moles} \times \text{molar mass (g mol}^{-1}\text{)} ]
So we need the molar mass of C₈H₁₈ first Which is the point..
Calculating the Molar Mass of C₈H₁₈
Carbon (C): 12.01 g mol⁻¹
Hydrogen (H): 1.008 g mol⁻¹
Octane has 8 carbons and 18 hydrogens:
[ \text{Molar mass} = (8 \times 12.01) + (18 \times 1.008) ]
Crunch the numbers:
- 8 × 12.01 = 96.08
- 18 × 1.008 = 18.144
Add them together:
[ 96.08 + 18.144 = 114 Small thing, real impact..
That’s the molar mass you’ll use.
Why It Matters / Why People Care
Knowing how to convert moles to grams isn’t just academic. Also, in a lab, you might need to weigh out exactly 5. Practically speaking, 90 mol of a reagent to run a reaction. In the automotive industry, fuel calculations rely on these conversions to predict performance. Even in everyday life, if you’re a hobbyist tinkering with homemade fuels or a student writing a lab report, you’ll hit this number. Skipping the molar mass step or using the wrong value can throw off the entire experiment It's one of those things that adds up..
How It Works (or How to Do It)
1. Grab the Atomic Weights
Use a periodic table. Practically speaking, carbon is 12. 01, hydrogen is 1.008. Some tables give rounded numbers, but for most lab work 12.01 and 1.008 are precise enough Easy to understand, harder to ignore..
2. Multiply by the Number of Atoms
For C₈H₁₈, multiply each atomic weight by its count:
- Carbon: 12.01 × 8
- Hydrogen: 1.008 × 18
3. Sum the Two Products
Add the two results to get the molar mass in grams per mole Simple as that..
4. Multiply by the Moles You Have
Now take the molar mass (114.224 g mol⁻¹) and multiply by 5.90 mol:
[ 114.224 \times 5.90 = 674.5456 \text{ g} ]
Round to a sensible number of significant figures. Since the molar mass was given to four significant figures and the mole count to three, round to three:
[ \boxed{675 \text{ g}} ]
That’s the mass of 5.90 mol of octane.
Common Mistakes / What Most People Get Wrong
-
Using the wrong molar mass – some people grab the average atomic weight (like 12.011 for carbon) and forget that the molar mass of a compound is the sum of exact atomic weights. The difference is tiny but can matter in precision work.
-
Mishandling significant figures – it’s easy to over‑specify the answer. The mole number (5.90) has three significant figures, so the final mass should match that.
-
Forgetting to convert units – if the molar mass is listed in kg mol⁻¹, you’ll need to convert to g mol⁻¹ before multiplying.
-
Mixing up molar mass and molecular weight – they’re the same thing, but some textbooks mislabel them, leading to confusion.
-
Dropping the decimal in the molar mass – 114 g mol⁻¹ instead of 114.224 g mol⁻¹ will give a noticeably wrong answer.
Practical Tips / What Actually Works
- Keep a small cheat‑sheet: jot down the molar masses of common reagents. For octane, just remember 114.2 g mol⁻¹.
- Use a calculator with a memory function so you can store 114.224 and reuse it for multiple problems.
- Practice rounding early. If you’re only interested in the nearest gram, you can round the molar mass to 114 g mol⁻¹ right away and get 114 × 5.90 = 673.6 g, then round to 674 g. That’s still within acceptable error for many lab settings.
- Double‑check with a different method. As an example, convert 5.90 mol to grams of carbon and hydrogen separately, then add. It’s a good sanity check.
- Use software or a spreadsheet for batch calculations. If you’re doing dozens of similar problems, a quick formula can save hours.
FAQ
Q1: Why is octane’s molar mass 114.224 g mol⁻¹ and not 114 g mol⁻¹?
A1: The slight difference comes from the exact atomic weights of carbon (12.01) and hydrogen (1.008). Rounding to 114 g mol⁻¹ is fine for rough estimates, but the full value is needed for precise work.
Q2: Can I use 12.011 for carbon instead of 12.01?
A2: Yes, 12.011 is the more precise value. It will change the final mass by less than a gram for 5.90 mol, but if you’re chasing high accuracy, use the more exact figure Easy to understand, harder to ignore..
Q3: What if the problem gives the mole value in millimoles?
A3: Convert millimoles to moles first (divide by 1,000), then follow the same steps. As an example, 5,900 mmol = 5.90 mol Most people skip this — try not to. Surprisingly effective..
Q4: Does temperature affect the mass calculation?
A4: No. Mass is invariant with temperature; only volume and density change. The mole‑to‑gram conversion is a purely stoichiometric calculation.
Q5: Is there a rule for rounding the final answer?
A5: Match the least number of significant figures in the input data. Here, 5.90 mol has three, so round the mass to three significant figures: 675 g.
Wrapping it up
You’ve just walked through the straightforward, yet essential, process of turning a mole count into a tangible weight. Whether you’re a student, a researcher, or a curious mind, the same steps apply to any compound. Remember the key: get the right molar mass, keep an eye on significant figures, and double‑check your work. Now you can tackle that next problem with confidence.
The beauty of this calculation lies in its universality. Once you master converting moles to grams for octane, you can apply the exact same method to water (18.In practice, 02 g/mol), glucose (180. Because of that, 16 g/mol), or any other substance. But the only thing that changes is the molar mass—everything else stays the same. This consistency is what makes stoichiometry such a powerful tool in chemistry.
As you continue your studies, you'll find that mole-to-gram conversions are just the beginning. They'll serve as the foundation for more complex calculations, such as determining limiting reactants, calculating theoretical yields, and balancing chemical equations in real-world experiments. The attention to detail you practice now—using precise atomic masses, tracking significant figures, and verifying your results—will pay dividends when reactions don't go exactly as planned and you need to troubleshoot where things went wrong Still holds up..
Don't be discouraged if you still feel unsure. Like any skill, proficiency comes with practice. Start with simple compounds, then gradually work your way up to molecules with multiple different elements. Keep that cheat-sheet handy, and don't hesitate to double-check your work using alternative methods. Over time, these calculations will become second nature, and you'll wonder why they ever seemed difficult.
So the next time you're faced with a chemistry problem asking how many grams are in a given number of moles, remember: it's simply moles multiplied by molar mass. Consider this: armed with this knowledge and the tips from this article, you're well-equipped to handle the challenge. Good luck, and happy calculating!
Practical Applications in Everyday Chemistry
Understanding mole-to-gram conversions opens doors to numerous real-world applications. In cooking, baking soda and baking powder quantities in recipes are carefully measured based on chemical reactions that depend on precise molar ratios. In medicine, dosage calculations for medications often rely on molecular weights and conversions between units. Environmental scientists use these same principles to measure pollutant concentrations in air and water, converting between moles and grams to determine safe exposure levels Simple as that..
Common Pitfalls to Avoid
Even experienced chemists occasionally stumble on a few recurring issues. One frequent mistake is forgetting to account for hydrates—compounds with water molecules incorporated into their crystal structure. So another trap is using outdated periodic table values; atomic masses are periodically refined as measurement techniques improve. Copper sulfate pentahydrate (CuSO₄·5H₂O), for instance, has a significantly higher molar mass than its anhydrous counterpart. Always verify you're working with current data, especially in academic or professional settings.
Tools of the Trade
Modern technology offers numerous resources to streamline these calculations. Digital periodic table apps provide instant molar masses for any element or compound. Spreadsheet templates can automate repetitive conversions, while chemistry-specific software handles complex molecules with multiple substituents. That said, understanding the fundamental mathematics remains essential—technology should augment your skills, not replace them.
Final Thoughts
The journey from moles to grams represents more than a mathematical exercise; it's a bridge between the abstract world of atomic-scale quantities and the tangible reality we measure in laboratories and industries. This conversion skill empowers you to translate theoretical equations into practical outcomes, whether you're synthesizing a new compound or analyzing environmental samples.
The official docs gloss over this. That's a mistake Most people skip this — try not to..
Chemistry is a language spoken through precision, patience, and persistence. Every calculation you master builds confidence for the next challenge. The mole-to-gram conversion is your foundation—build upon it wisely, and the possibilities are endless.